H5N1 in 2012: The Year in Review
Laidback Al, Senior Moderator, www.FluTrackers.com
January 1, 2013
Abstract ? In 2012, 32 human cases of H5N1 were reported from around the world, the lowest number of reported human cases since the World Health Organization (WHO) began reporting H5N1 cases in 2003. In 2012, six countries reported cases, Bangladesh, Cambodia, China, Egypt, Indonesia, and Viet Nam. More than 60% of the reported cases died, a slight increase in the fatality rate over 2011. Although there were only 32 cases reported in 2012, five are associated with two clusters. The source of infection for all of the cases has been reported as exposure to sick and dying poultry.
Also, in 2012, controversy over H5N1 laboratory experimentation erupted, several genetic studies were published, and seroprevalence results were evaluated. Although fewer cases were reported in 2012 than preceding years, an H5N1 pandemic is still a potential worldwide public health threat.
Background
Influenza A(H5N1) (often referred to ?bird flu? or avian influenza or HPAI) is a novel influenza virus. It was first detected in humans in Hong Kong in 1997. Since 2003, the World Health Organization (WHO) has been monitoring human outbreaks of H5N1, because of its potential to become a pandemic virus. The current WHO report on Human infection with avian influenza A(H5N1) viruses and associated animal health events was updated on December 17, 2012. Through December 31, 2012, WHO has officially reported a total of 610 confirmed human cases of H5N1. The most recent WHO time line of significant events associated with the H5N1 virus was last updated on June 15, 2012 (link).
The following summary of human cases from 2012 is generally organized according to the outlined presented last year in ?A Summary of Human H5N1 Cases in 2011?(FT link). Additional discussions of H5N1 topics from 2012 are also presented in this review.
1.0 Summary of Human H5N1 Cases in 2012
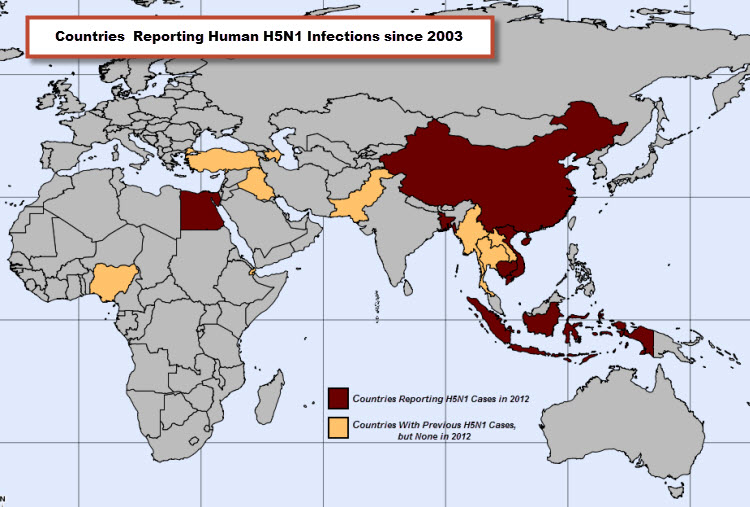
The tabulation for WHO-confirmed H5N1 cases in 2012 is 32, including the two December-reported cases, one each from Egypt and Indonesia. Compared to 2011, the number of worldwide H5N1 cases declined in 2012 by about 50%. Since 2003, 15 countries from around the world have reported human H5N1 cases to WHO. In 2012, 6 countries reported human cases of H5N1, Bangladesh, Cambodia, China, Egypt, Indonesia and Viet Nam. Each of these countries has previously reported human cases.
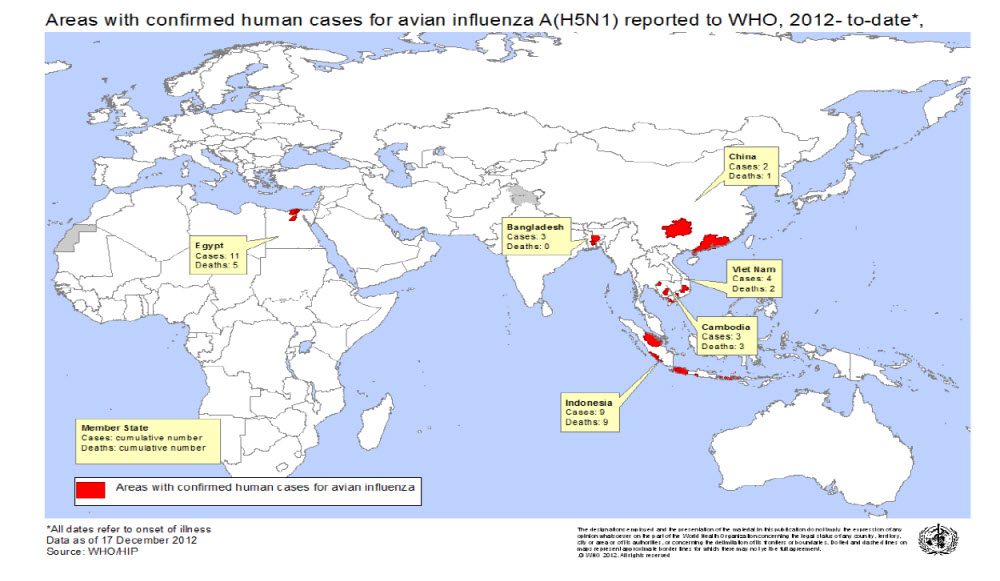
WHO map of countries (Administrative level 1) with human H5N1 cases in2012.
All countries reporting human H5N1 cases since 2003.
Twenty of the 32 cases reported in 2012 cases occurred in two countries, Egypt and Indonesia. These two countries, in total, have reported almost 60% of all cases reported worldwide since 2003. Indonesia continues to lead the world in cumulative number of reported human H5N1 cases with 192. Egypt is second with 169 reported cases.
1.1 H5N1 Case-Fatality Rate in 2012
Of the 32 cases in 2012, 20 died, with an overall case-fatality rate (CFR) of 62.5% for 2012. All nine reported cases in Indonesia and all three cases from Cambodia were reported as fatalities, a CFR of 100% in 2012 in each of these two countries. Although Cambodia has only reported 21 cases since 2005, the last time Cambodia reported a survivor from an H5N1 infection was in 2009. Although there is inter-year variability, the overall CFR for the 610 WHO reported cases is 59%.
1.2 Demographics of H5N1 Cases in 2012
In the past several years, females outnumbered males among WHO reported cases. In 2012, the ratio of males to females was exactly 1:1. Sixteen of the reported cases were female, and 16 were male. Overall, females represent about 53% of all of the WHO reported cases where sex was noted. In 2012, a higher percentage of females died (68%), 11 of 16, while only 9 of the 16 males (56%) died. These rates are similar to overall fatality rate by sex for all cases. Among identified females in the WHO case list, about 64% of them died. This contrasts with identified males; only about 55% died.
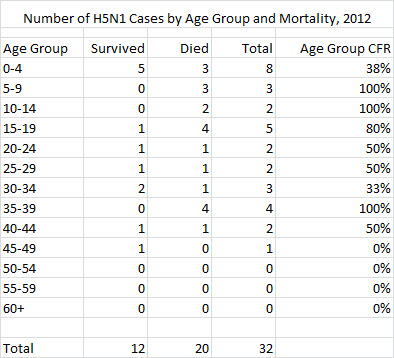
The ages of H5N1 cases from 2012 range from 1 to 45 years old, with a median age of 18. The median age of infection in 2012 is higher than 2011, when it was 11. Children under 4 years represent about 25% of all H5N1 infections in 2012, and notably, these cases have a higher survivorship rate than most other age groups.
Table of 2012 H5N1 infections by age group.
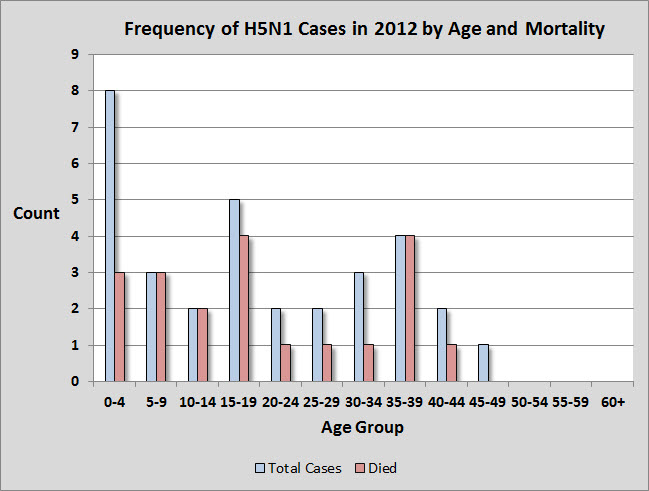
1.3 H5N1 Clusters in 2012
In 2012 only two H5N1 clusters were identified, one from Indonesia and one from Bangladesh. A cluster is defined as two or more cases of confirmed, probable, or suspected Influenza A(H5N1) infections with onset of illness occurring within the same two-week period and who are in the same geographical area and/or are epidemiologically linked (WHO link).
1.3.1 Indonesia Cluster, December 2011-January 2012
A 23 year-old male from Sunter Agung district, Tanjung Priok, North Jarkarta, apparently contracted H5N1 from a sick pigeon. He experienced onset on December 31, 2011, was hospitalized on January 4, 2012, and died on January 7. A sister of the male, a 5 year-old girl, was hospitalized on January 7, 2012, and later died on January 16. The young girl was reported to have contact with the sick pigeon as well (FT link). Both cases were reported by WHO.
1.3.2 Bangladesh Cluster, February-March 2012
In late February and early March 2012, three workers at a live bird, wet market were confirmed with H5N1 infections in southern Dhaka City, Bangladesh. The three workers, all males ranging in age from 18 to 40, apparently presented with symptoms including fever, sore throat, and cough. Each tested positive for H5N1. There are no indications of treatment but all three recovered (IEDCR link, IEDCR link). The Director of IEDCR, Prof. Mahmudur Rhaman, in a media report, implied that these infections were a result of clade 2.2 (link). Infection from infected poultry is presumed for all three of these cases. All three of these cases were reported by WHO.
1.3.3 Study of H5N1 Clusters, Indonesia
In 2012, PloS ONE published an article prepared by Aditama and colleagues, entitled Avian Influenza H5N1 Transmission in Households, Indonesia.This publication is important because it is the first comprehensive effort to understand infection and transmission among H5N1 cases in Indonesia. The authors investigated 113 isolated outbreaks (represented by one individual) and 26 cluster outbreaks that occurred between 2005 and 2009. Their study involved 177 cases that occurred between July 2005 and July 2009. Their analysis demonstrated that even with the clustering of some of the cases, only limited human-to-human transmission was noted among some of the cluster cases.
There is a discrepancy between the number of cases used in this study (177) and the number of cases reported by WHO during the same time period. The total number of WHO reported cases from Indonesia between 2005 and 2009 is 162, 15 cases less than the number cited in this study. It would be beneficial if a line list of all cases used within this study were published and made available online.
2.0 H5N1 Sequence Analysis, Egypt
In 2012, Younan and colleagues presented a study of genetic and antigenic diversity of subtype H5N1 viruses isolated from humans in Egypt between 2007?2011 in Microevolution of Highly Pathogenic Avian Influenza A(H5N1) Viruses Isolated from Humans, Egypt, 2007?2011.
Based on the analysis of the complete genomes of viruses isolated from 59 humans in Egypt obtained between 2007?2011, the authors report the following findings. The recent subtype H5N1 viruses isolated from human infections in Egypt originated from poultry and evolved from a single genotype introduced into Egypt in 2005/2006. Of the human infections during 2009?2011, 95% were caused by viruses from a single phylogenetic group, clade 2.2.1-C. There is no evidence of subsequent reassortment with new subtype H5N1 virus genes introduced into Egypt or resident LPAI viruses. Importantly, the recent 2011 isolates were antigenically closely related to the proposed WHO candidate vaccine virus, A/Egypt/N03072/2010, indicating a good antigenic match between currently circulating strains and the proposed vaccine.
3.0 H5N1 Mutations and Transmissibility
From late 2011 to early 2012, there was substantial scientific debate and numerous media reports about the bioethical implications of laboratory experimentation with laboratory modified H5 isolates by the Fouchier team (link) and Kawaoka team (link). Dr. Ron Fouchier, leading a team from the Erasmus Center in the Netherlands, demonstrated that several substitutions are all that is necessary for the H5N1 virus to acquire a capacity for airborne transmission (link). During this same time, Dr. Yoshihiro Kawaoka heading a team of researchers from the University and Wisconsin conducted a similar study of droplet transmission with an H5 HA reassortant virus (link). The Kawaoka report states
In light the these laboratory experiments, a recent PLoS Pathogen article takes on added significance. In Egyptian H5N1 Influenza Viruses?Cause for Concern? - Gabriele Neumann, Catherine A. Macken, Alexander I. Karasin, Ron A. M. Fouchier, Yoshihiro Kawaoka,the authors,which include both Fouchier and Kawaoka, state
4.0 H5N1 Seroprevalance Analysis
Arising from the bioethical discussion of the Fouchier and Kawaoka studies were a series of exchanges regarding the interpretation of H5N1 seroprevalance data in the Proceedings of the National Academy of Sciences and Mbio (link, link, link, link) and the journal Science (link, link, link).
Underlying the initial exchanges in PNAS was an assumption that the suppression of Fouchier?s and Kawaoka?s laboratory research was prompted by the high H5N1 CFR based on WHO data. Palese and Wang (link) suggest fear mongering and cite several seroprevalence studies indicating subclinical H5N1 infections would lower the CFR for H5N1.
In March, Wang and colleagues (link) presented a meta-analysis of H5N1 serostudies and concluded that the rate of H5N1 infection in the study populations ranges from 1-2%. A series of arguments and counter arguments ensued regarding the adequacy of serological testing procedures, the nature of the sampled populations (e.g., poultry workers versus a random sample of the local population), restrictions of testing to countries with sporadic or endemic H5N1, and the appropriateness of accepting or rejecting a particular study in the meta-analysis.
The serological studies demonstrate quite clearly that a small fraction of some of the tested populations show evidence of previous subclinical or asymptomatic H5N1 infections; human cases of H5N1 infections that are not included in the official case count by WHO. Because the serological studies were drawn from a different sampling universe than the WHO line list of cases, it is not possible to align or correlate the statistical data from the seroprevalence studies with the WHO data.
The WHO line list of H5N1 cases is an independent data set. It is not a representative data set in a statistical sense and was not drawn from the same population as the seroprevalence studies. The 610 WHO cases cannot be accepted as a sample or subset of any larger identifiable population. Therefore, the results of the serological studies cannot be used to provide a denominator to estimate an accurate CFR for H5N1 infections.
While the seroprevalence data cannot directly be used to predict the virulence and CFR of an H5N1 pandemic in the future, further research on serological studies is necessary. A recent study, entitled A Serological Survey of Antibodies to H5, H7 and H9 Avian Influenza Viruses amongst the Duck-Related Workers in Beijing, China, was published after the meta-analysis cited above. The authors of this study from China examined the seroprevalence of antibodies against H5, H7, and H9 of 1741 duck-related workers in Beijing, China. A small number had antibody against H9, but none were seropositive for H5 or H7. Based on the meta-analysis cited above, it is an unexpected result that no seropositive H5 cases were discovered among the duck workers.
5.0 H5N1 Reassortment
While the discussions in 2012 focused on laboratory created H5 isolates, there was little discussion regarding the potential for H5N1 reassortant strains to develop in the wild. The 2009 H1N1 pandemic is a reminder that an H5N1 virus could reassort with other circulating influenza viruses in humans and other mammals. Any one of the cases reported by WHO or the individuals testing positive in the serostudies could have been a host for reassortment.
6.0 Conclusion
Despite the fact that humans have been sporadically infected with H5N1 for more than 15 years and that the reported number of human cases have reached a 10-year low, the world should not be lulled into a false sense of security and complacency. H5N1 is still an emerging infectious zoonotic disease and is still a likely candidate for a pandemic virus. Together, the proliferation of H5N1 clades in the past decade, the laboratory demonstration of transmissibility of H5 viruses, circulating H5N1 viruses in Egypt with transmissibility potential, and our poor understanding of a CFR for an H5N1 pandemic, all demonstrate the need for continued surveillance of human and animal outbreaks of H5N1, and an increased emphasis on the full range of laboratory research to understand the H5N1 virus.
Notes
1. The data and information used here have been derived from numerous publicly available online sources including WHO, various ministries of health reports, and other internet media reports. For some individual cases, details such as age, sex, specific residence, etc. are lacking. Also news media reports sometime provide conflicting data about individual cases.The basic data and information presented here is believed to be reasonably accurate.The information presented here is based on data reported through December 31, 2012. Because WHO uses the date of onset to assign a case to particular year, there could be some cases that will be retrospectively assigned to 2012 at a later date which will affect the data presented here. Any errors or omissions are mine, however.
Acknowledgements and Disclaimer
I would like to thank all of the posters and moderators at FluTrackers.com, internet bloggers, and other internet forum members for their efforts at online tracking of H5N1 and other emerging infectious diseases. Thanks are also due to open source journals and researchers who post full copies of their papers. The opinions expressed here are my own and do not necessarily represent those of FluTrackers.com.
<!--[if gte mso 9]><xml> <w:WordDocument> <w:View>Normal</w:View> <w:Zoom>0</w:Zoom> <w:TrackMoves/> <w:TrackFormatting/> <w:PunctuationKerning/> <w:ValidateAgainstSchemas/> <w:SaveIfXMLInvalid>false</w:SaveIfXMLInvalid> <w:IgnoreMixedContent>false</w:IgnoreMixedContent> <w:AlwaysShowPlaceholderText>false</w:AlwaysShowPlaceholderText> <w oNotPromoteQF/> <w:LidThemeOther>EN-US</w:LidThemeOther> <w:LidThemeAsian>X-NONE</w:LidThemeAsian> <w:LidThemeComplexScript>KHM</w:LidThemeComplexScript> <w:Compatibility> <w:BreakWrappedTables/> <w:SnapToGridInCell/> <w:WrapTextWithPunct/> <w:UseAsianBreakRules/> <w
oNotPromoteQF/> <w:LidThemeOther>EN-US</w:LidThemeOther> <w:LidThemeAsian>X-NONE</w:LidThemeAsian> <w:LidThemeComplexScript>KHM</w:LidThemeComplexScript> <w:Compatibility> <w:BreakWrappedTables/> <w:SnapToGridInCell/> <w:WrapTextWithPunct/> <w:UseAsianBreakRules/> <w ontGrowAutofit/> <w:SplitPgBreakAndParaMark/> <w:EnableOpenTypeKerning/> <w
ontGrowAutofit/> <w:SplitPgBreakAndParaMark/> <w:EnableOpenTypeKerning/> <w ontFlipMirrorIndents/> <w:OverrideTableStyleHps/> </w:Compatibility> <m:mathPr> <m:mathFont m:val="Cambria Math"/> <m:brkBin m:val="before"/> <m:brkBinSub m:val="--"/> <m:smallFrac m:val="off"/> <m:dispDef/> <m:lMargin m:val="0"/> <m:rMargin m:val="0"/> <m:defJc m:val="centerGroup"/> <m:wrapIndent m:val="1440"/> <m:intLim m:val="subSup"/> <m:naryLim m:val="undOvr"/> </m:mathPr></w:WordDocument> </xml><![endif]--><!--[if gte mso 9]><xml> <w:LatentStyles DefLockedState="false" DefUnhideWhenUsed="true" DefSemiHidden="true" DefQFormat="false" DefPriority="99" LatentStyleCount="267"> <w:LsdException Locked="false" Priority="0" SemiHidden="false" UnhideWhenUsed="false" QFormat="true" Name="Normal"/> <w:LsdException Locked="false" Priority="9" SemiHidden="false" UnhideWhenUsed="false" QFormat="true" Name="heading 1"/> <w:LsdException Locked="false" Priority="9" QFormat="true" Name="heading 2"/> <w:LsdException Locked="false" Priority="9" QFormat="true" Name="heading 3"/> <w:LsdException Locked="false" Priority="9" QFormat="true" Name="heading 4"/> <w:LsdException Locked="false" Priority="9" QFormat="true" Name="heading 5"/> <w:LsdException Locked="false" Priority="9" QFormat="true" Name="heading 6"/> <w:LsdException Locked="false" Priority="9" QFormat="true" Name="heading 7"/> <w:LsdException Locked="false" Priority="9" QFormat="true" Name="heading 8"/> <w:LsdException Locked="false" Priority="9" QFormat="true" Name="heading 9"/> <w:LsdException Locked="false" Priority="39" Name="toc 1"/> <w:LsdException Locked="false" Priority="39" Name="toc 2"/> <w:LsdException Locked="false" Priority="39" Name="toc 3"/> <w:LsdException Locked="false" Priority="39" Name="toc 4"/> <w:LsdException Locked="false" Priority="39" Name="toc 5"/> <w:LsdException Locked="false" Priority="39" Name="toc 6"/> <w:LsdException Locked="false" Priority="39" Name="toc 7"/> <w:LsdException Locked="false" Priority="39" Name="toc 8"/> <w:LsdException Locked="false" Priority="39" Name="toc 9"/> <w:LsdException Locked="false" Priority="35" QFormat="true" Name="caption"/> <w:LsdException Locked="false" Priority="10" SemiHidden="false" UnhideWhenUsed="false" QFormat="true" Name="Title"/> <w:LsdException Locked="false" Priority="1" Name="Default Paragraph Font"/> <w:LsdException Locked="false" Priority="11" SemiHidden="false" UnhideWhenUsed="false" QFormat="true" Name="Subtitle"/> <w:LsdException Locked="false" Priority="22" SemiHidden="false" UnhideWhenUsed="false" QFormat="true" Name="Strong"/> <w:LsdException Locked="false" Priority="20" SemiHidden="false" UnhideWhenUsed="false" QFormat="true" Name="Emphasis"/> <w:LsdException Locked="false" Priority="59" SemiHidden="false" UnhideWhenUsed="false" Name="Table Grid"/> <w:LsdException Locked="false" UnhideWhenUsed="false" Name="Placeholder Text"/> <w:LsdException Locked="false" Priority="1" SemiHidden="false" UnhideWhenUsed="false" QFormat="true" Name="No Spacing"/> <w:LsdException Locked="false" Priority="60" SemiHidden="false" UnhideWhenUsed="false" Name="Light Shading"/> <w:LsdException Locked="false" Priority="61" SemiHidden="false" UnhideWhenUsed="false" Name="Light List"/> <w:LsdException Locked="false" Priority="62" SemiHidden="false" UnhideWhenUsed="false" Name="Light Grid"/> <w:LsdException Locked="false" Priority="63" SemiHidden="false" UnhideWhenUsed="false" Name="Medium Shading 1"/> <w:LsdException Locked="false" Priority="64" SemiHidden="false" UnhideWhenUsed="false" Name="Medium Shading 2"/> <w:LsdException Locked="false" Priority="65" SemiHidden="false" UnhideWhenUsed="false" Name="Medium List 1"/> <w:LsdException Locked="false" Priority="66" SemiHidden="false" UnhideWhenUsed="false" Name="Medium List 2"/> <w:LsdException Locked="false" Priority="67" SemiHidden="false" UnhideWhenUsed="false" Name="Medium Grid 1"/> <w:LsdException Locked="false" Priority="68" SemiHidden="false" UnhideWhenUsed="false" Name="Medium Grid 2"/> <w:LsdException Locked="false" Priority="69" SemiHidden="false" UnhideWhenUsed="false" Name="Medium Grid 3"/> <w:LsdException Locked="false" Priority="70" SemiHidden="false" UnhideWhenUsed="false" Name="Dark List"/> <w:LsdException Locked="false" Priority="71" SemiHidden="false" UnhideWhenUsed="false" Name="Colorful Shading"/> <w:LsdException Locked="false" Priority="72" SemiHidden="false" UnhideWhenUsed="false" Name="Colorful List"/> <w:LsdException Locked="false" Priority="73" SemiHidden="false" UnhideWhenUsed="false" Name="Colorful Grid"/> <w:LsdException Locked="false" Priority="60" SemiHidden="false" UnhideWhenUsed="false" Name="Light Shading Accent 1"/> <w:LsdException Locked="false" Priority="61" SemiHidden="false" UnhideWhenUsed="false" Name="Light List Accent 1"/> <w:LsdException Locked="false" Priority="62" SemiHidden="false" UnhideWhenUsed="false" Name="Light Grid Accent 1"/> <w:LsdException Locked="false" Priority="63" SemiHidden="false" UnhideWhenUsed="false" Name="Medium Shading 1 Accent 1"/> <w:LsdException Locked="false" Priority="64" SemiHidden="false" UnhideWhenUsed="false" Name="Medium Shading 2 Accent 1"/> <w:LsdException Locked="false" Priority="65" SemiHidden="false" UnhideWhenUsed="false" Name="Medium List 1 Accent 1"/> <w:LsdException Locked="false" UnhideWhenUsed="false" Name="Revision"/> <w:LsdException Locked="false" Priority="34" SemiHidden="false" UnhideWhenUsed="false" QFormat="true" Name="List Paragraph"/> <w:LsdException Locked="false" Priority="29" SemiHidden="false" UnhideWhenUsed="false" QFormat="true" Name="Quote"/> <w:LsdException Locked="false" Priority="30" SemiHidden="false" UnhideWhenUsed="false" QFormat="true" Name="Intense Quote"/> <w:LsdException Locked="false" Priority="66" SemiHidden="false" UnhideWhenUsed="false" Name="Medium List 2 Accent 1"/> <w:LsdException Locked="false" Priority="67" SemiHidden="false" UnhideWhenUsed="false" Name="Medium Grid 1 Accent 1"/> <w:LsdException Locked="false" Priority="68" SemiHidden="false" UnhideWhenUsed="false" Name="Medium Grid 2 Accent 1"/> <w:LsdException Locked="false" Priority="69" SemiHidden="false" UnhideWhenUsed="false" Name="Medium Grid 3 Accent 1"/> <w:LsdException Locked="false" Priority="70" SemiHidden="false" UnhideWhenUsed="false" Name="Dark List Accent 1"/> <w:LsdException Locked="false" Priority="71" SemiHidden="false" UnhideWhenUsed="false" Name="Colorful Shading Accent 1"/> <w:LsdException Locked="false" Priority="72" SemiHidden="false" UnhideWhenUsed="false" Name="Colorful List Accent 1"/> <w:LsdException Locked="false" Priority="73" SemiHidden="false" UnhideWhenUsed="false" Name="Colorful Grid Accent 1"/> <w:LsdException Locked="false" Priority="60" SemiHidden="false" UnhideWhenUsed="false" Name="Light Shading Accent 2"/> <w:LsdException Locked="false" Priority="61" SemiHidden="false" UnhideWhenUsed="false" Name="Light List Accent 2"/> <w:LsdException Locked="false" Priority="62" SemiHidden="false" UnhideWhenUsed="false" Name="Light Grid Accent 2"/> <w:LsdException Locked="false" Priority="63" SemiHidden="false" UnhideWhenUsed="false" Name="Medium Shading 1 Accent 2"/> <w:LsdException Locked="false" Priority="64" SemiHidden="false" UnhideWhenUsed="false" Name="Medium Shading 2 Accent 2"/> <w:LsdException Locked="false" Priority="65" SemiHidden="false" UnhideWhenUsed="false" Name="Medium List 1 Accent 2"/> <w:LsdException Locked="false" Priority="66" SemiHidden="false" UnhideWhenUsed="false" Name="Medium List 2 Accent 2"/> <w:LsdException Locked="false" Priority="67" SemiHidden="false" UnhideWhenUsed="false" Name="Medium Grid 1 Accent 2"/> <w:LsdException Locked="false" Priority="68" SemiHidden="false" UnhideWhenUsed="false" Name="Medium Grid 2 Accent 2"/> <w:LsdException Locked="false" Priority="69" SemiHidden="false" UnhideWhenUsed="false" Name="Medium Grid 3 Accent 2"/> <w:LsdException Locked="false" Priority="70" SemiHidden="false" UnhideWhenUsed="false" Name="Dark List Accent 2"/> <w:LsdException Locked="false" Priority="71" SemiHidden="false" UnhideWhenUsed="false" Name="Colorful Shading Accent 2"/> <w:LsdException Locked="false" Priority="72" SemiHidden="false" UnhideWhenUsed="false" Name="Colorful List Accent 2"/> <w:LsdException Locked="false" Priority="73" SemiHidden="false" UnhideWhenUsed="false" Name="Colorful Grid Accent 2"/> <w:LsdException Locked="false" Priority="60" SemiHidden="false" UnhideWhenUsed="false" Name="Light Shading Accent 3"/> <w:LsdException Locked="false" Priority="61" SemiHidden="false" UnhideWhenUsed="false" Name="Light List Accent 3"/> <w:LsdException Locked="false" Priority="62" SemiHidden="false" UnhideWhenUsed="false" Name="Light Grid Accent 3"/> <w:LsdException Locked="false" Priority="63" SemiHidden="false" UnhideWhenUsed="false" Name="Medium Shading 1 Accent 3"/> <w:LsdException Locked="false" Priority="64" SemiHidden="false" UnhideWhenUsed="false" Name="Medium Shading 2 Accent 3"/> <w:LsdException Locked="false" Priority="65" SemiHidden="false" UnhideWhenUsed="false" Name="Medium List 1 Accent 3"/> <w:LsdException Locked="false" Priority="66" SemiHidden="false" UnhideWhenUsed="false" Name="Medium List 2 Accent 3"/> <w:LsdException Locked="false" Priority="67" SemiHidden="false" UnhideWhenUsed="false" Name="Medium Grid 1 Accent 3"/> <w:LsdException Locked="false" Priority="68" SemiHidden="false" UnhideWhenUsed="false" Name="Medium Grid 2 Accent 3"/> <w:LsdException Locked="false" Priority="69" SemiHidden="false" UnhideWhenUsed="false" Name="Medium Grid 3 Accent 3"/> <w:LsdException Locked="false" Priority="70" SemiHidden="false" UnhideWhenUsed="false" Name="Dark List Accent 3"/> <w:LsdException Locked="false" Priority="71" SemiHidden="false" UnhideWhenUsed="false" Name="Colorful Shading Accent 3"/> <w:LsdException Locked="false" Priority="72" SemiHidden="false" UnhideWhenUsed="false" Name="Colorful List Accent 3"/> <w:LsdException Locked="false" Priority="73" SemiHidden="false" UnhideWhenUsed="false" Name="Colorful Grid Accent 3"/> <w:LsdException Locked="false" Priority="60" SemiHidden="false" UnhideWhenUsed="false" Name="Light Shading Accent 4"/> <w:LsdException Locked="false" Priority="61" SemiHidden="false" UnhideWhenUsed="false" Name="Light List Accent 4"/> <w:LsdException Locked="false" Priority="62" SemiHidden="false" UnhideWhenUsed="false" Name="Light Grid Accent 4"/> <w:LsdException Locked="false" Priority="63" SemiHidden="false" UnhideWhenUsed="false" Name="Medium Shading 1 Accent 4"/> <w:LsdException Locked="false" Priority="64" SemiHidden="false" UnhideWhenUsed="false" Name="Medium Shading 2 Accent 4"/> <w:LsdException Locked="false" Priority="65" SemiHidden="false" UnhideWhenUsed="false" Name="Medium List 1 Accent 4"/> <w:LsdException Locked="false" Priority="66" SemiHidden="false" UnhideWhenUsed="false" Name="Medium List 2 Accent 4"/> <w:LsdException Locked="false" Priority="67" SemiHidden="false" UnhideWhenUsed="false" Name="Medium Grid 1 Accent 4"/> <w:LsdException Locked="false" Priority="68" SemiHidden="false" UnhideWhenUsed="false" Name="Medium Grid 2 Accent 4"/> <w:LsdException Locked="false" Priority="69" SemiHidden="false" UnhideWhenUsed="false" Name="Medium Grid 3 Accent 4"/> <w:LsdException Locked="false" Priority="70" SemiHidden="false" UnhideWhenUsed="false" Name="Dark List Accent 4"/> <w:LsdException Locked="false" Priority="71" SemiHidden="false" UnhideWhenUsed="false" Name="Colorful Shading Accent 4"/> <w:LsdException Locked="false" Priority="72" SemiHidden="false" UnhideWhenUsed="false" Name="Colorful List Accent 4"/> <w:LsdException Locked="false" Priority="73" SemiHidden="false" UnhideWhenUsed="false" Name="Colorful Grid Accent 4"/> <w:LsdException Locked="false" Priority="60" SemiHidden="false" UnhideWhenUsed="false" Name="Light Shading Accent 5"/> <w:LsdException Locked="false" Priority="61" SemiHidden="false" UnhideWhenUsed="false" Name="Light List Accent 5"/> <w:LsdException Locked="false" Priority="62" SemiHidden="false" UnhideWhenUsed="false" Name="Light Grid Accent 5"/> <w:LsdException Locked="false" Priority="63" SemiHidden="false" UnhideWhenUsed="false" Name="Medium Shading 1 Accent 5"/> <w:LsdException Locked="false" Priority="64" SemiHidden="false" UnhideWhenUsed="false" Name="Medium Shading 2 Accent 5"/> <w:LsdException Locked="false" Priority="65" SemiHidden="false" UnhideWhenUsed="false" Name="Medium List 1 Accent 5"/> <w:LsdException Locked="false" Priority="66" SemiHidden="false" UnhideWhenUsed="false" Name="Medium List 2 Accent 5"/> <w:LsdException Locked="false" Priority="67" SemiHidden="false" UnhideWhenUsed="false" Name="Medium Grid 1 Accent 5"/> <w:LsdException Locked="false" Priority="68" SemiHidden="false" UnhideWhenUsed="false" Name="Medium Grid 2 Accent 5"/> <w:LsdException Locked="false" Priority="69" SemiHidden="false" UnhideWhenUsed="false" Name="Medium Grid 3 Accent 5"/> <w:LsdException Locked="false" Priority="70" SemiHidden="false" UnhideWhenUsed="false" Name="Dark List Accent 5"/> <w:LsdException Locked="false" Priority="71" SemiHidden="false" UnhideWhenUsed="false" Name="Colorful Shading Accent 5"/> <w:LsdException Locked="false" Priority="72" SemiHidden="false" UnhideWhenUsed="false" Name="Colorful List Accent 5"/> <w:LsdException Locked="false" Priority="73" SemiHidden="false" UnhideWhenUsed="false" Name="Colorful Grid Accent 5"/> <w:LsdException Locked="false" Priority="60" SemiHidden="false" UnhideWhenUsed="false" Name="Light Shading Accent 6"/> <w:LsdException Locked="false" Priority="61" SemiHidden="false" UnhideWhenUsed="false" Name="Light List Accent 6"/> <w:LsdException Locked="false" Priority="62" SemiHidden="false" UnhideWhenUsed="false" Name="Light Grid Accent 6"/> <w:LsdException Locked="false" Priority="63" SemiHidden="false" UnhideWhenUsed="false" Name="Medium Shading 1 Accent 6"/> <w:LsdException Locked="false" Priority="64" SemiHidden="false" UnhideWhenUsed="false" Name="Medium Shading 2 Accent 6"/> <w:LsdException Locked="false" Priority="65" SemiHidden="false" UnhideWhenUsed="false" Name="Medium List 1 Accent 6"/> <w:LsdException Locked="false" Priority="66" SemiHidden="false" UnhideWhenUsed="false" Name="Medium List 2 Accent 6"/> <w:LsdException Locked="false" Priority="67" SemiHidden="false" UnhideWhenUsed="false" Name="Medium Grid 1 Accent 6"/> <w:LsdException Locked="false" Priority="68" SemiHidden="false" UnhideWhenUsed="false" Name="Medium Grid 2 Accent 6"/> <w:LsdException Locked="false" Priority="69" SemiHidden="false" UnhideWhenUsed="false" Name="Medium Grid 3 Accent 6"/> <w:LsdException Locked="false" Priority="70" SemiHidden="false" UnhideWhenUsed="false" Name="Dark List Accent 6"/> <w:LsdException Locked="false" Priority="71" SemiHidden="false" UnhideWhenUsed="false" Name="Colorful Shading Accent 6"/> <w:LsdException Locked="false" Priority="72" SemiHidden="false" UnhideWhenUsed="false" Name="Colorful List Accent 6"/> <w:LsdException Locked="false" Priority="73" SemiHidden="false" UnhideWhenUsed="false" Name="Colorful Grid Accent 6"/> <w:LsdException Locked="false" Priority="19" SemiHidden="false" UnhideWhenUsed="false" QFormat="true" Name="Subtle Emphasis"/> <w:LsdException Locked="false" Priority="21" SemiHidden="false" UnhideWhenUsed="false" QFormat="true" Name="Intense Emphasis"/> <w:LsdException Locked="false" Priority="31" SemiHidden="false" UnhideWhenUsed="false" QFormat="true" Name="Subtle Reference"/> <w:LsdException Locked="false" Priority="32" SemiHidden="false" UnhideWhenUsed="false" QFormat="true" Name="Intense Reference"/> <w:LsdException Locked="false" Priority="33" SemiHidden="false" UnhideWhenUsed="false" QFormat="true" Name="Book Title"/> <w:LsdException Locked="false" Priority="37" Name="Bibliography"/> <w:LsdException Locked="false" Priority="39" QFormat="true" Name="TOC Heading"/> </w:LatentStyles> </xml><![endif]--><!--[if gte mso 10]> <style> /* Style Definitions */ table.MsoNormalTable {mso-style-name:"Table Normal"; mso-tstyle-rowband-size:0; mso-tstyle-colband-size:0; mso-style-noshow:yes; mso-style-priority:99; mso-style-parent:""; mso-padding-alt:0in 5.4pt 0in 5.4pt; mso-para-margin:0in; mso-para-margin-bottom:.0001pt; mso-pagination:widow-orphan; font-size:10.0pt; font-family:"Calibri","sans-serif";} </style> <![endif]--><object style="position:absolute;z-index:1000" type="application/x-dgnria" id="plugin0" height="0" width="0">
ontFlipMirrorIndents/> <w:OverrideTableStyleHps/> </w:Compatibility> <m:mathPr> <m:mathFont m:val="Cambria Math"/> <m:brkBin m:val="before"/> <m:brkBinSub m:val="--"/> <m:smallFrac m:val="off"/> <m:dispDef/> <m:lMargin m:val="0"/> <m:rMargin m:val="0"/> <m:defJc m:val="centerGroup"/> <m:wrapIndent m:val="1440"/> <m:intLim m:val="subSup"/> <m:naryLim m:val="undOvr"/> </m:mathPr></w:WordDocument> </xml><![endif]--><!--[if gte mso 9]><xml> <w:LatentStyles DefLockedState="false" DefUnhideWhenUsed="true" DefSemiHidden="true" DefQFormat="false" DefPriority="99" LatentStyleCount="267"> <w:LsdException Locked="false" Priority="0" SemiHidden="false" UnhideWhenUsed="false" QFormat="true" Name="Normal"/> <w:LsdException Locked="false" Priority="9" SemiHidden="false" UnhideWhenUsed="false" QFormat="true" Name="heading 1"/> <w:LsdException Locked="false" Priority="9" QFormat="true" Name="heading 2"/> <w:LsdException Locked="false" Priority="9" QFormat="true" Name="heading 3"/> <w:LsdException Locked="false" Priority="9" QFormat="true" Name="heading 4"/> <w:LsdException Locked="false" Priority="9" QFormat="true" Name="heading 5"/> <w:LsdException Locked="false" Priority="9" QFormat="true" Name="heading 6"/> <w:LsdException Locked="false" Priority="9" QFormat="true" Name="heading 7"/> <w:LsdException Locked="false" Priority="9" QFormat="true" Name="heading 8"/> <w:LsdException Locked="false" Priority="9" QFormat="true" Name="heading 9"/> <w:LsdException Locked="false" Priority="39" Name="toc 1"/> <w:LsdException Locked="false" Priority="39" Name="toc 2"/> <w:LsdException Locked="false" Priority="39" Name="toc 3"/> <w:LsdException Locked="false" Priority="39" Name="toc 4"/> <w:LsdException Locked="false" Priority="39" Name="toc 5"/> <w:LsdException Locked="false" Priority="39" Name="toc 6"/> <w:LsdException Locked="false" Priority="39" Name="toc 7"/> <w:LsdException Locked="false" Priority="39" Name="toc 8"/> <w:LsdException Locked="false" Priority="39" Name="toc 9"/> <w:LsdException Locked="false" Priority="35" QFormat="true" Name="caption"/> <w:LsdException Locked="false" Priority="10" SemiHidden="false" UnhideWhenUsed="false" QFormat="true" Name="Title"/> <w:LsdException Locked="false" Priority="1" Name="Default Paragraph Font"/> <w:LsdException Locked="false" Priority="11" SemiHidden="false" UnhideWhenUsed="false" QFormat="true" Name="Subtitle"/> <w:LsdException Locked="false" Priority="22" SemiHidden="false" UnhideWhenUsed="false" QFormat="true" Name="Strong"/> <w:LsdException Locked="false" Priority="20" SemiHidden="false" UnhideWhenUsed="false" QFormat="true" Name="Emphasis"/> <w:LsdException Locked="false" Priority="59" SemiHidden="false" UnhideWhenUsed="false" Name="Table Grid"/> <w:LsdException Locked="false" UnhideWhenUsed="false" Name="Placeholder Text"/> <w:LsdException Locked="false" Priority="1" SemiHidden="false" UnhideWhenUsed="false" QFormat="true" Name="No Spacing"/> <w:LsdException Locked="false" Priority="60" SemiHidden="false" UnhideWhenUsed="false" Name="Light Shading"/> <w:LsdException Locked="false" Priority="61" SemiHidden="false" UnhideWhenUsed="false" Name="Light List"/> <w:LsdException Locked="false" Priority="62" SemiHidden="false" UnhideWhenUsed="false" Name="Light Grid"/> <w:LsdException Locked="false" Priority="63" SemiHidden="false" UnhideWhenUsed="false" Name="Medium Shading 1"/> <w:LsdException Locked="false" Priority="64" SemiHidden="false" UnhideWhenUsed="false" Name="Medium Shading 2"/> <w:LsdException Locked="false" Priority="65" SemiHidden="false" UnhideWhenUsed="false" Name="Medium List 1"/> <w:LsdException Locked="false" Priority="66" SemiHidden="false" UnhideWhenUsed="false" Name="Medium List 2"/> <w:LsdException Locked="false" Priority="67" SemiHidden="false" UnhideWhenUsed="false" Name="Medium Grid 1"/> <w:LsdException Locked="false" Priority="68" SemiHidden="false" UnhideWhenUsed="false" Name="Medium Grid 2"/> <w:LsdException Locked="false" Priority="69" SemiHidden="false" UnhideWhenUsed="false" Name="Medium Grid 3"/> <w:LsdException Locked="false" Priority="70" SemiHidden="false" UnhideWhenUsed="false" Name="Dark List"/> <w:LsdException Locked="false" Priority="71" SemiHidden="false" UnhideWhenUsed="false" Name="Colorful Shading"/> <w:LsdException Locked="false" Priority="72" SemiHidden="false" UnhideWhenUsed="false" Name="Colorful List"/> <w:LsdException Locked="false" Priority="73" SemiHidden="false" UnhideWhenUsed="false" Name="Colorful Grid"/> <w:LsdException Locked="false" Priority="60" SemiHidden="false" UnhideWhenUsed="false" Name="Light Shading Accent 1"/> <w:LsdException Locked="false" Priority="61" SemiHidden="false" UnhideWhenUsed="false" Name="Light List Accent 1"/> <w:LsdException Locked="false" Priority="62" SemiHidden="false" UnhideWhenUsed="false" Name="Light Grid Accent 1"/> <w:LsdException Locked="false" Priority="63" SemiHidden="false" UnhideWhenUsed="false" Name="Medium Shading 1 Accent 1"/> <w:LsdException Locked="false" Priority="64" SemiHidden="false" UnhideWhenUsed="false" Name="Medium Shading 2 Accent 1"/> <w:LsdException Locked="false" Priority="65" SemiHidden="false" UnhideWhenUsed="false" Name="Medium List 1 Accent 1"/> <w:LsdException Locked="false" UnhideWhenUsed="false" Name="Revision"/> <w:LsdException Locked="false" Priority="34" SemiHidden="false" UnhideWhenUsed="false" QFormat="true" Name="List Paragraph"/> <w:LsdException Locked="false" Priority="29" SemiHidden="false" UnhideWhenUsed="false" QFormat="true" Name="Quote"/> <w:LsdException Locked="false" Priority="30" SemiHidden="false" UnhideWhenUsed="false" QFormat="true" Name="Intense Quote"/> <w:LsdException Locked="false" Priority="66" SemiHidden="false" UnhideWhenUsed="false" Name="Medium List 2 Accent 1"/> <w:LsdException Locked="false" Priority="67" SemiHidden="false" UnhideWhenUsed="false" Name="Medium Grid 1 Accent 1"/> <w:LsdException Locked="false" Priority="68" SemiHidden="false" UnhideWhenUsed="false" Name="Medium Grid 2 Accent 1"/> <w:LsdException Locked="false" Priority="69" SemiHidden="false" UnhideWhenUsed="false" Name="Medium Grid 3 Accent 1"/> <w:LsdException Locked="false" Priority="70" SemiHidden="false" UnhideWhenUsed="false" Name="Dark List Accent 1"/> <w:LsdException Locked="false" Priority="71" SemiHidden="false" UnhideWhenUsed="false" Name="Colorful Shading Accent 1"/> <w:LsdException Locked="false" Priority="72" SemiHidden="false" UnhideWhenUsed="false" Name="Colorful List Accent 1"/> <w:LsdException Locked="false" Priority="73" SemiHidden="false" UnhideWhenUsed="false" Name="Colorful Grid Accent 1"/> <w:LsdException Locked="false" Priority="60" SemiHidden="false" UnhideWhenUsed="false" Name="Light Shading Accent 2"/> <w:LsdException Locked="false" Priority="61" SemiHidden="false" UnhideWhenUsed="false" Name="Light List Accent 2"/> <w:LsdException Locked="false" Priority="62" SemiHidden="false" UnhideWhenUsed="false" Name="Light Grid Accent 2"/> <w:LsdException Locked="false" Priority="63" SemiHidden="false" UnhideWhenUsed="false" Name="Medium Shading 1 Accent 2"/> <w:LsdException Locked="false" Priority="64" SemiHidden="false" UnhideWhenUsed="false" Name="Medium Shading 2 Accent 2"/> <w:LsdException Locked="false" Priority="65" SemiHidden="false" UnhideWhenUsed="false" Name="Medium List 1 Accent 2"/> <w:LsdException Locked="false" Priority="66" SemiHidden="false" UnhideWhenUsed="false" Name="Medium List 2 Accent 2"/> <w:LsdException Locked="false" Priority="67" SemiHidden="false" UnhideWhenUsed="false" Name="Medium Grid 1 Accent 2"/> <w:LsdException Locked="false" Priority="68" SemiHidden="false" UnhideWhenUsed="false" Name="Medium Grid 2 Accent 2"/> <w:LsdException Locked="false" Priority="69" SemiHidden="false" UnhideWhenUsed="false" Name="Medium Grid 3 Accent 2"/> <w:LsdException Locked="false" Priority="70" SemiHidden="false" UnhideWhenUsed="false" Name="Dark List Accent 2"/> <w:LsdException Locked="false" Priority="71" SemiHidden="false" UnhideWhenUsed="false" Name="Colorful Shading Accent 2"/> <w:LsdException Locked="false" Priority="72" SemiHidden="false" UnhideWhenUsed="false" Name="Colorful List Accent 2"/> <w:LsdException Locked="false" Priority="73" SemiHidden="false" UnhideWhenUsed="false" Name="Colorful Grid Accent 2"/> <w:LsdException Locked="false" Priority="60" SemiHidden="false" UnhideWhenUsed="false" Name="Light Shading Accent 3"/> <w:LsdException Locked="false" Priority="61" SemiHidden="false" UnhideWhenUsed="false" Name="Light List Accent 3"/> <w:LsdException Locked="false" Priority="62" SemiHidden="false" UnhideWhenUsed="false" Name="Light Grid Accent 3"/> <w:LsdException Locked="false" Priority="63" SemiHidden="false" UnhideWhenUsed="false" Name="Medium Shading 1 Accent 3"/> <w:LsdException Locked="false" Priority="64" SemiHidden="false" UnhideWhenUsed="false" Name="Medium Shading 2 Accent 3"/> <w:LsdException Locked="false" Priority="65" SemiHidden="false" UnhideWhenUsed="false" Name="Medium List 1 Accent 3"/> <w:LsdException Locked="false" Priority="66" SemiHidden="false" UnhideWhenUsed="false" Name="Medium List 2 Accent 3"/> <w:LsdException Locked="false" Priority="67" SemiHidden="false" UnhideWhenUsed="false" Name="Medium Grid 1 Accent 3"/> <w:LsdException Locked="false" Priority="68" SemiHidden="false" UnhideWhenUsed="false" Name="Medium Grid 2 Accent 3"/> <w:LsdException Locked="false" Priority="69" SemiHidden="false" UnhideWhenUsed="false" Name="Medium Grid 3 Accent 3"/> <w:LsdException Locked="false" Priority="70" SemiHidden="false" UnhideWhenUsed="false" Name="Dark List Accent 3"/> <w:LsdException Locked="false" Priority="71" SemiHidden="false" UnhideWhenUsed="false" Name="Colorful Shading Accent 3"/> <w:LsdException Locked="false" Priority="72" SemiHidden="false" UnhideWhenUsed="false" Name="Colorful List Accent 3"/> <w:LsdException Locked="false" Priority="73" SemiHidden="false" UnhideWhenUsed="false" Name="Colorful Grid Accent 3"/> <w:LsdException Locked="false" Priority="60" SemiHidden="false" UnhideWhenUsed="false" Name="Light Shading Accent 4"/> <w:LsdException Locked="false" Priority="61" SemiHidden="false" UnhideWhenUsed="false" Name="Light List Accent 4"/> <w:LsdException Locked="false" Priority="62" SemiHidden="false" UnhideWhenUsed="false" Name="Light Grid Accent 4"/> <w:LsdException Locked="false" Priority="63" SemiHidden="false" UnhideWhenUsed="false" Name="Medium Shading 1 Accent 4"/> <w:LsdException Locked="false" Priority="64" SemiHidden="false" UnhideWhenUsed="false" Name="Medium Shading 2 Accent 4"/> <w:LsdException Locked="false" Priority="65" SemiHidden="false" UnhideWhenUsed="false" Name="Medium List 1 Accent 4"/> <w:LsdException Locked="false" Priority="66" SemiHidden="false" UnhideWhenUsed="false" Name="Medium List 2 Accent 4"/> <w:LsdException Locked="false" Priority="67" SemiHidden="false" UnhideWhenUsed="false" Name="Medium Grid 1 Accent 4"/> <w:LsdException Locked="false" Priority="68" SemiHidden="false" UnhideWhenUsed="false" Name="Medium Grid 2 Accent 4"/> <w:LsdException Locked="false" Priority="69" SemiHidden="false" UnhideWhenUsed="false" Name="Medium Grid 3 Accent 4"/> <w:LsdException Locked="false" Priority="70" SemiHidden="false" UnhideWhenUsed="false" Name="Dark List Accent 4"/> <w:LsdException Locked="false" Priority="71" SemiHidden="false" UnhideWhenUsed="false" Name="Colorful Shading Accent 4"/> <w:LsdException Locked="false" Priority="72" SemiHidden="false" UnhideWhenUsed="false" Name="Colorful List Accent 4"/> <w:LsdException Locked="false" Priority="73" SemiHidden="false" UnhideWhenUsed="false" Name="Colorful Grid Accent 4"/> <w:LsdException Locked="false" Priority="60" SemiHidden="false" UnhideWhenUsed="false" Name="Light Shading Accent 5"/> <w:LsdException Locked="false" Priority="61" SemiHidden="false" UnhideWhenUsed="false" Name="Light List Accent 5"/> <w:LsdException Locked="false" Priority="62" SemiHidden="false" UnhideWhenUsed="false" Name="Light Grid Accent 5"/> <w:LsdException Locked="false" Priority="63" SemiHidden="false" UnhideWhenUsed="false" Name="Medium Shading 1 Accent 5"/> <w:LsdException Locked="false" Priority="64" SemiHidden="false" UnhideWhenUsed="false" Name="Medium Shading 2 Accent 5"/> <w:LsdException Locked="false" Priority="65" SemiHidden="false" UnhideWhenUsed="false" Name="Medium List 1 Accent 5"/> <w:LsdException Locked="false" Priority="66" SemiHidden="false" UnhideWhenUsed="false" Name="Medium List 2 Accent 5"/> <w:LsdException Locked="false" Priority="67" SemiHidden="false" UnhideWhenUsed="false" Name="Medium Grid 1 Accent 5"/> <w:LsdException Locked="false" Priority="68" SemiHidden="false" UnhideWhenUsed="false" Name="Medium Grid 2 Accent 5"/> <w:LsdException Locked="false" Priority="69" SemiHidden="false" UnhideWhenUsed="false" Name="Medium Grid 3 Accent 5"/> <w:LsdException Locked="false" Priority="70" SemiHidden="false" UnhideWhenUsed="false" Name="Dark List Accent 5"/> <w:LsdException Locked="false" Priority="71" SemiHidden="false" UnhideWhenUsed="false" Name="Colorful Shading Accent 5"/> <w:LsdException Locked="false" Priority="72" SemiHidden="false" UnhideWhenUsed="false" Name="Colorful List Accent 5"/> <w:LsdException Locked="false" Priority="73" SemiHidden="false" UnhideWhenUsed="false" Name="Colorful Grid Accent 5"/> <w:LsdException Locked="false" Priority="60" SemiHidden="false" UnhideWhenUsed="false" Name="Light Shading Accent 6"/> <w:LsdException Locked="false" Priority="61" SemiHidden="false" UnhideWhenUsed="false" Name="Light List Accent 6"/> <w:LsdException Locked="false" Priority="62" SemiHidden="false" UnhideWhenUsed="false" Name="Light Grid Accent 6"/> <w:LsdException Locked="false" Priority="63" SemiHidden="false" UnhideWhenUsed="false" Name="Medium Shading 1 Accent 6"/> <w:LsdException Locked="false" Priority="64" SemiHidden="false" UnhideWhenUsed="false" Name="Medium Shading 2 Accent 6"/> <w:LsdException Locked="false" Priority="65" SemiHidden="false" UnhideWhenUsed="false" Name="Medium List 1 Accent 6"/> <w:LsdException Locked="false" Priority="66" SemiHidden="false" UnhideWhenUsed="false" Name="Medium List 2 Accent 6"/> <w:LsdException Locked="false" Priority="67" SemiHidden="false" UnhideWhenUsed="false" Name="Medium Grid 1 Accent 6"/> <w:LsdException Locked="false" Priority="68" SemiHidden="false" UnhideWhenUsed="false" Name="Medium Grid 2 Accent 6"/> <w:LsdException Locked="false" Priority="69" SemiHidden="false" UnhideWhenUsed="false" Name="Medium Grid 3 Accent 6"/> <w:LsdException Locked="false" Priority="70" SemiHidden="false" UnhideWhenUsed="false" Name="Dark List Accent 6"/> <w:LsdException Locked="false" Priority="71" SemiHidden="false" UnhideWhenUsed="false" Name="Colorful Shading Accent 6"/> <w:LsdException Locked="false" Priority="72" SemiHidden="false" UnhideWhenUsed="false" Name="Colorful List Accent 6"/> <w:LsdException Locked="false" Priority="73" SemiHidden="false" UnhideWhenUsed="false" Name="Colorful Grid Accent 6"/> <w:LsdException Locked="false" Priority="19" SemiHidden="false" UnhideWhenUsed="false" QFormat="true" Name="Subtle Emphasis"/> <w:LsdException Locked="false" Priority="21" SemiHidden="false" UnhideWhenUsed="false" QFormat="true" Name="Intense Emphasis"/> <w:LsdException Locked="false" Priority="31" SemiHidden="false" UnhideWhenUsed="false" QFormat="true" Name="Subtle Reference"/> <w:LsdException Locked="false" Priority="32" SemiHidden="false" UnhideWhenUsed="false" QFormat="true" Name="Intense Reference"/> <w:LsdException Locked="false" Priority="33" SemiHidden="false" UnhideWhenUsed="false" QFormat="true" Name="Book Title"/> <w:LsdException Locked="false" Priority="37" Name="Bibliography"/> <w:LsdException Locked="false" Priority="39" QFormat="true" Name="TOC Heading"/> </w:LatentStyles> </xml><![endif]--><!--[if gte mso 10]> <style> /* Style Definitions */ table.MsoNormalTable {mso-style-name:"Table Normal"; mso-tstyle-rowband-size:0; mso-tstyle-colband-size:0; mso-style-noshow:yes; mso-style-priority:99; mso-style-parent:""; mso-padding-alt:0in 5.4pt 0in 5.4pt; mso-para-margin:0in; mso-para-margin-bottom:.0001pt; mso-pagination:widow-orphan; font-size:10.0pt; font-family:"Calibri","sans-serif";} </style> <![endif]--><object style="position:absolute;z-index:1000" type="application/x-dgnria" id="plugin0" height="0" width="0">
</object>
Laidback Al, Senior Moderator, www.FluTrackers.com
January 1, 2013
Abstract ? In 2012, 32 human cases of H5N1 were reported from around the world, the lowest number of reported human cases since the World Health Organization (WHO) began reporting H5N1 cases in 2003. In 2012, six countries reported cases, Bangladesh, Cambodia, China, Egypt, Indonesia, and Viet Nam. More than 60% of the reported cases died, a slight increase in the fatality rate over 2011. Although there were only 32 cases reported in 2012, five are associated with two clusters. The source of infection for all of the cases has been reported as exposure to sick and dying poultry.
Also, in 2012, controversy over H5N1 laboratory experimentation erupted, several genetic studies were published, and seroprevalence results were evaluated. Although fewer cases were reported in 2012 than preceding years, an H5N1 pandemic is still a potential worldwide public health threat.
Background
Influenza A(H5N1) (often referred to ?bird flu? or avian influenza or HPAI) is a novel influenza virus. It was first detected in humans in Hong Kong in 1997. Since 2003, the World Health Organization (WHO) has been monitoring human outbreaks of H5N1, because of its potential to become a pandemic virus. The current WHO report on Human infection with avian influenza A(H5N1) viruses and associated animal health events was updated on December 17, 2012. Through December 31, 2012, WHO has officially reported a total of 610 confirmed human cases of H5N1. The most recent WHO time line of significant events associated with the H5N1 virus was last updated on June 15, 2012 (link).
The following summary of human cases from 2012 is generally organized according to the outlined presented last year in ?A Summary of Human H5N1 Cases in 2011?(FT link). Additional discussions of H5N1 topics from 2012 are also presented in this review.
1.0 Summary of Human H5N1 Cases in 2012
The tabulation for WHO-confirmed H5N1 cases in 2012 is 32, including the two December-reported cases, one each from Egypt and Indonesia. Compared to 2011, the number of worldwide H5N1 cases declined in 2012 by about 50%. Since 2003, 15 countries from around the world have reported human H5N1 cases to WHO. In 2012, 6 countries reported human cases of H5N1, Bangladesh, Cambodia, China, Egypt, Indonesia and Viet Nam. Each of these countries has previously reported human cases.
WHO map of countries (Administrative level 1) with human H5N1 cases in2012.
All countries reporting human H5N1 cases since 2003.
Twenty of the 32 cases reported in 2012 cases occurred in two countries, Egypt and Indonesia. These two countries, in total, have reported almost 60% of all cases reported worldwide since 2003. Indonesia continues to lead the world in cumulative number of reported human H5N1 cases with 192. Egypt is second with 169 reported cases.
1.1 H5N1 Case-Fatality Rate in 2012
Of the 32 cases in 2012, 20 died, with an overall case-fatality rate (CFR) of 62.5% for 2012. All nine reported cases in Indonesia and all three cases from Cambodia were reported as fatalities, a CFR of 100% in 2012 in each of these two countries. Although Cambodia has only reported 21 cases since 2005, the last time Cambodia reported a survivor from an H5N1 infection was in 2009. Although there is inter-year variability, the overall CFR for the 610 WHO reported cases is 59%.
1.2 Demographics of H5N1 Cases in 2012
In the past several years, females outnumbered males among WHO reported cases. In 2012, the ratio of males to females was exactly 1:1. Sixteen of the reported cases were female, and 16 were male. Overall, females represent about 53% of all of the WHO reported cases where sex was noted. In 2012, a higher percentage of females died (68%), 11 of 16, while only 9 of the 16 males (56%) died. These rates are similar to overall fatality rate by sex for all cases. Among identified females in the WHO case list, about 64% of them died. This contrasts with identified males; only about 55% died.
The ages of H5N1 cases from 2012 range from 1 to 45 years old, with a median age of 18. The median age of infection in 2012 is higher than 2011, when it was 11. Children under 4 years represent about 25% of all H5N1 infections in 2012, and notably, these cases have a higher survivorship rate than most other age groups.
Table of 2012 H5N1 infections by age group.
1.3 H5N1 Clusters in 2012
In 2012 only two H5N1 clusters were identified, one from Indonesia and one from Bangladesh. A cluster is defined as two or more cases of confirmed, probable, or suspected Influenza A(H5N1) infections with onset of illness occurring within the same two-week period and who are in the same geographical area and/or are epidemiologically linked (WHO link).
1.3.1 Indonesia Cluster, December 2011-January 2012
A 23 year-old male from Sunter Agung district, Tanjung Priok, North Jarkarta, apparently contracted H5N1 from a sick pigeon. He experienced onset on December 31, 2011, was hospitalized on January 4, 2012, and died on January 7. A sister of the male, a 5 year-old girl, was hospitalized on January 7, 2012, and later died on January 16. The young girl was reported to have contact with the sick pigeon as well (FT link). Both cases were reported by WHO.
1.3.2 Bangladesh Cluster, February-March 2012
In late February and early March 2012, three workers at a live bird, wet market were confirmed with H5N1 infections in southern Dhaka City, Bangladesh. The three workers, all males ranging in age from 18 to 40, apparently presented with symptoms including fever, sore throat, and cough. Each tested positive for H5N1. There are no indications of treatment but all three recovered (IEDCR link, IEDCR link). The Director of IEDCR, Prof. Mahmudur Rhaman, in a media report, implied that these infections were a result of clade 2.2 (link). Infection from infected poultry is presumed for all three of these cases. All three of these cases were reported by WHO.
1.3.3 Study of H5N1 Clusters, Indonesia
In 2012, PloS ONE published an article prepared by Aditama and colleagues, entitled Avian Influenza H5N1 Transmission in Households, Indonesia.This publication is important because it is the first comprehensive effort to understand infection and transmission among H5N1 cases in Indonesia. The authors investigated 113 isolated outbreaks (represented by one individual) and 26 cluster outbreaks that occurred between 2005 and 2009. Their study involved 177 cases that occurred between July 2005 and July 2009. Their analysis demonstrated that even with the clustering of some of the cases, only limited human-to-human transmission was noted among some of the cluster cases.
There is a discrepancy between the number of cases used in this study (177) and the number of cases reported by WHO during the same time period. The total number of WHO reported cases from Indonesia between 2005 and 2009 is 162, 15 cases less than the number cited in this study. It would be beneficial if a line list of all cases used within this study were published and made available online.
2.0 H5N1 Sequence Analysis, Egypt
In 2012, Younan and colleagues presented a study of genetic and antigenic diversity of subtype H5N1 viruses isolated from humans in Egypt between 2007?2011 in Microevolution of Highly Pathogenic Avian Influenza A(H5N1) Viruses Isolated from Humans, Egypt, 2007?2011.
Based on the analysis of the complete genomes of viruses isolated from 59 humans in Egypt obtained between 2007?2011, the authors report the following findings. The recent subtype H5N1 viruses isolated from human infections in Egypt originated from poultry and evolved from a single genotype introduced into Egypt in 2005/2006. Of the human infections during 2009?2011, 95% were caused by viruses from a single phylogenetic group, clade 2.2.1-C. There is no evidence of subsequent reassortment with new subtype H5N1 virus genes introduced into Egypt or resident LPAI viruses. Importantly, the recent 2011 isolates were antigenically closely related to the proposed WHO candidate vaccine virus, A/Egypt/N03072/2010, indicating a good antigenic match between currently circulating strains and the proposed vaccine.
3.0 H5N1 Mutations and Transmissibility
From late 2011 to early 2012, there was substantial scientific debate and numerous media reports about the bioethical implications of laboratory experimentation with laboratory modified H5 isolates by the Fouchier team (link) and Kawaoka team (link). Dr. Ron Fouchier, leading a team from the Erasmus Center in the Netherlands, demonstrated that several substitutions are all that is necessary for the H5N1 virus to acquire a capacity for airborne transmission (link). During this same time, Dr. Yoshihiro Kawaoka heading a team of researchers from the University and Wisconsin conducted a similar study of droplet transmission with an H5 HA reassortant virus (link). The Kawaoka report states
?Three of the residues identified here (N224, Q226 and T318) have been strictly conserved among H5 HA proteins isolated since 2003. . . . One of the four mutations we identified in our transmissible virus, the N158D mutation, results in loss of a glycosylation site. Many H5N1 viruses isolated in the Middle East, Africa, Asia and Europe do not have this glycosylation site. Therefore, only three nucleotide changes are needed for the HA of these viruses to support efficient transmission in ferrets. In addition, the H5N1 viruses circulating in these geographic areas also possess a glutamic-acid-to-lysine mutation at position 627 in the PB2 protein, which promotes viral replication in certain mammals, including humans. Therefore, these viruses may be several steps closer to those capable of efficient transmission in humans and are of concern.?
?Our database searches identified two H5N1 viruses that encode HA-220K and have lost the HA154?156 glycosylation site (A/muscovy duck/Vietnam/NCVD-11/2007; A/duck/Egypt/10185SS/2010), indicating that only two additional mutations are needed to create variants with the ?transmissibility features? identified in the Kawaoka study.? They conclude ?Because the outbreak of H5N1 viruses in Egypt is extensive, Egyptian H5N1 viruses may, therefore, present a far greater pandemic risk than H5N1 viruses circulating in other countries.?
Arising from the bioethical discussion of the Fouchier and Kawaoka studies were a series of exchanges regarding the interpretation of H5N1 seroprevalance data in the Proceedings of the National Academy of Sciences and Mbio (link, link, link, link) and the journal Science (link, link, link).
Underlying the initial exchanges in PNAS was an assumption that the suppression of Fouchier?s and Kawaoka?s laboratory research was prompted by the high H5N1 CFR based on WHO data. Palese and Wang (link) suggest fear mongering and cite several seroprevalence studies indicating subclinical H5N1 infections would lower the CFR for H5N1.
In March, Wang and colleagues (link) presented a meta-analysis of H5N1 serostudies and concluded that the rate of H5N1 infection in the study populations ranges from 1-2%. A series of arguments and counter arguments ensued regarding the adequacy of serological testing procedures, the nature of the sampled populations (e.g., poultry workers versus a random sample of the local population), restrictions of testing to countries with sporadic or endemic H5N1, and the appropriateness of accepting or rejecting a particular study in the meta-analysis.
The serological studies demonstrate quite clearly that a small fraction of some of the tested populations show evidence of previous subclinical or asymptomatic H5N1 infections; human cases of H5N1 infections that are not included in the official case count by WHO. Because the serological studies were drawn from a different sampling universe than the WHO line list of cases, it is not possible to align or correlate the statistical data from the seroprevalence studies with the WHO data.
The WHO line list of H5N1 cases is an independent data set. It is not a representative data set in a statistical sense and was not drawn from the same population as the seroprevalence studies. The 610 WHO cases cannot be accepted as a sample or subset of any larger identifiable population. Therefore, the results of the serological studies cannot be used to provide a denominator to estimate an accurate CFR for H5N1 infections.
While the seroprevalence data cannot directly be used to predict the virulence and CFR of an H5N1 pandemic in the future, further research on serological studies is necessary. A recent study, entitled A Serological Survey of Antibodies to H5, H7 and H9 Avian Influenza Viruses amongst the Duck-Related Workers in Beijing, China, was published after the meta-analysis cited above. The authors of this study from China examined the seroprevalence of antibodies against H5, H7, and H9 of 1741 duck-related workers in Beijing, China. A small number had antibody against H9, but none were seropositive for H5 or H7. Based on the meta-analysis cited above, it is an unexpected result that no seropositive H5 cases were discovered among the duck workers.
5.0 H5N1 Reassortment
While the discussions in 2012 focused on laboratory created H5 isolates, there was little discussion regarding the potential for H5N1 reassortant strains to develop in the wild. The 2009 H1N1 pandemic is a reminder that an H5N1 virus could reassort with other circulating influenza viruses in humans and other mammals. Any one of the cases reported by WHO or the individuals testing positive in the serostudies could have been a host for reassortment.
6.0 Conclusion
Despite the fact that humans have been sporadically infected with H5N1 for more than 15 years and that the reported number of human cases have reached a 10-year low, the world should not be lulled into a false sense of security and complacency. H5N1 is still an emerging infectious zoonotic disease and is still a likely candidate for a pandemic virus. Together, the proliferation of H5N1 clades in the past decade, the laboratory demonstration of transmissibility of H5 viruses, circulating H5N1 viruses in Egypt with transmissibility potential, and our poor understanding of a CFR for an H5N1 pandemic, all demonstrate the need for continued surveillance of human and animal outbreaks of H5N1, and an increased emphasis on the full range of laboratory research to understand the H5N1 virus.
Notes
1. The data and information used here have been derived from numerous publicly available online sources including WHO, various ministries of health reports, and other internet media reports. For some individual cases, details such as age, sex, specific residence, etc. are lacking. Also news media reports sometime provide conflicting data about individual cases.The basic data and information presented here is believed to be reasonably accurate.The information presented here is based on data reported through December 31, 2012. Because WHO uses the date of onset to assign a case to particular year, there could be some cases that will be retrospectively assigned to 2012 at a later date which will affect the data presented here. Any errors or omissions are mine, however.
Acknowledgements and Disclaimer
I would like to thank all of the posters and moderators at FluTrackers.com, internet bloggers, and other internet forum members for their efforts at online tracking of H5N1 and other emerging infectious diseases. Thanks are also due to open source journals and researchers who post full copies of their papers. The opinions expressed here are my own and do not necessarily represent those of FluTrackers.com.
<!--[if gte mso 9]><xml> <w:WordDocument> <w:View>Normal</w:View> <w:Zoom>0</w:Zoom> <w:TrackMoves/> <w:TrackFormatting/> <w:PunctuationKerning/> <w:ValidateAgainstSchemas/> <w:SaveIfXMLInvalid>false</w:SaveIfXMLInvalid> <w:IgnoreMixedContent>false</w:IgnoreMixedContent> <w:AlwaysShowPlaceholderText>false</w:AlwaysShowPlaceholderText> <w
</object>